Bioorthogonal Neurotransmitters
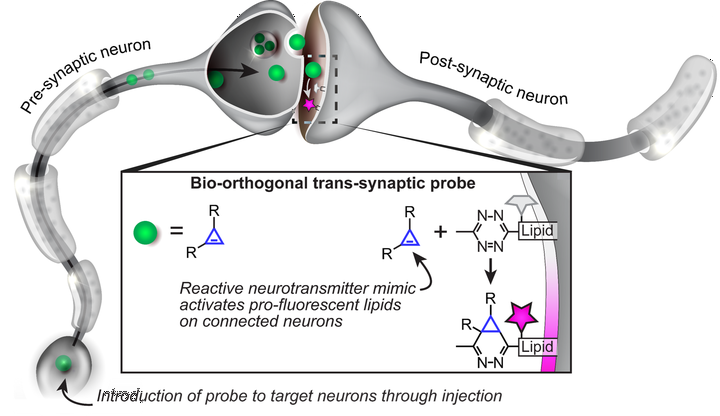
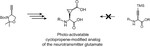
Substituted cyclopropenes serve as compact biorthogonal appendages that enable analysis of biomolecules in complex systems. Neurotransmitters, a chemically diverse group of biomolecules that control neuron excitation and inhibition, are not among the systems that have been studied using biorthogonal chemistry. Here we describe:
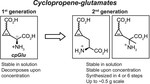
1. Synthesis of cyclopropene-containing analogs of the excitatory amino acid neurotransmitter glutamate2. Synthesis of a photoactivatable version of cyclopropene-glutamate3. Bioorthogonal reaction of such cyclopropene-glutamates with tetrazines and tetrazolesThis strategy has the potential to permit deployment of cyclopropene-modified glutamate as a bioorthogonal probe of the neurotransmitter glutamate in vivo with spatiotemporal precision.