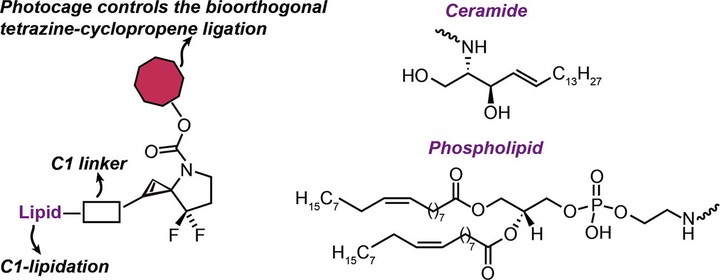
Lipidated Spirocyclopropyls
We have developed an inexpensive, catalyst-free and multigram-scale synthesis of cyclic enamines with exocyclic double bonds, for four to seven-membered rings. This strategy is more conducive to scale up, permissive of functionalization around the cyclic system, and less sensitive to the nature of the N-protecting group than previously-described methods for cyclic enamine synthesis.
Further, we explore application of these enamines to the synthesis of highly-strained spirocyclic cyclopropenes. We also made lipidated versions of these cyclopropenes that can serve as useful bioorthogonal reagents for imaging cell membranes via pro-fluorescent tetrazines.